|
How did Goldstein prove the existence of protons in an atom? As a result, he reasoned that the particles in the cathode ray were negative. Thomson found that the path taken by the cathode ray could be bent towards a positive metal plate, and away from a negative metal plate. Why did JJ Thomson conclude that the particles were negatively charged? Rutherford’s gold foil experiment showed that the atom is mostly empty space with a tiny, dense, positively-charged nucleus. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. What did JJ Thomson’s cathode ray tube experiment prove? 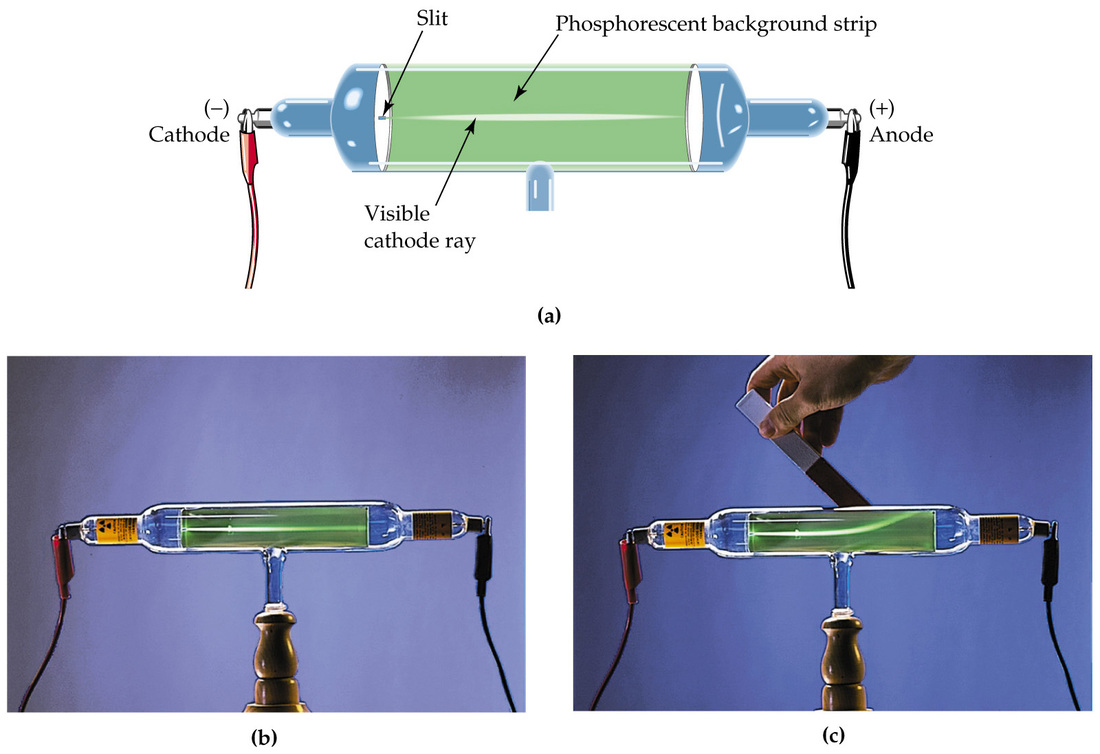
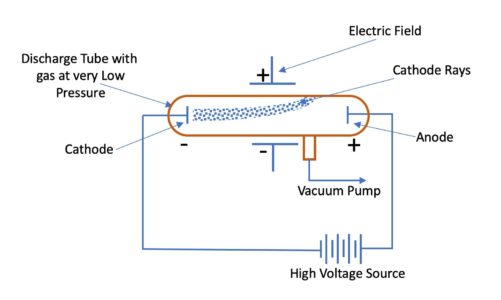
Thomson discovered that the mass of the particles in cathode rays was 1800 times lighter than hydrogen, the lightest element. Johann Hittorf discovered cathode rays in 1869 using a Crookes tube and noting shadows cast on the glowing wall of the tube opposite of the cathode. What did the cathode ray experiment prove? 
Accordingly, he called his particles electrons. Thomson’s discovery established the particulate nature of electricity. With both magnetic and electric deflections observed, it was clear that cathode rays were negatively charged particles. Thomson’s plum pudding model of the atom had negatively-charged electrons embedded within a positively-charged “soup.” Did the cathode ray have positive or negative charge? What experiment proved electrons carry a negative charge? Millikan discovered that there is a fundamental electric charge-the charge of an electron. Thomson’s cathode ray tube showed that atoms contain small, negatively charged particles called electrons. What evidence supports the existence of negatively charged particles?Īlthough no one has actually seen the inside of an atom, experiments have demonstrated much about atomic structure. The negative electrons represented the raisins in the pudding and the dough contained the positive charge. He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. How did Thomson’s evidence prove that electrons are negatively charged? Thomson used the cathode ray tube to determine that atoms had small negatively charged particles inside of them, which he called “electrons.” Electrons were first discovered as the constituents of cathode rays. How does the cathode ray tube experiment proved the existence of electrons?Ĭathode rays carry electronic currents through the tube. He observed that cathode rays were deflected by a magnetic field in the same manner as a wire carrying an electric current, which was known to be negatively charged. In order to determine if the cathode ray consisted of charged particles, Thomson used magnets and charged plates to deflect the cathode ray. 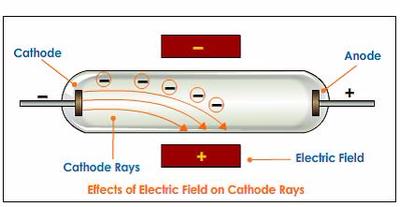
How it was proved that cathode rays carry negative charge?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
